BioAcyl Corp

BioAcyl Corp |
 |
| Resource type: Journal Article DOI: 10.1038/s41591-021-01329-2 ID no. (ISBN etc.): 1546-170X BibTeX citation key: Stephenson2021 View all bibliographic details |
Categories: BioAcyl Corp Subcategories: Asymptomatic COVID-19 Creators: Botting, others, Reynolds, Stephenson Collection: Nat. Med. |
Views: 4/317
|
| Abstract |
|
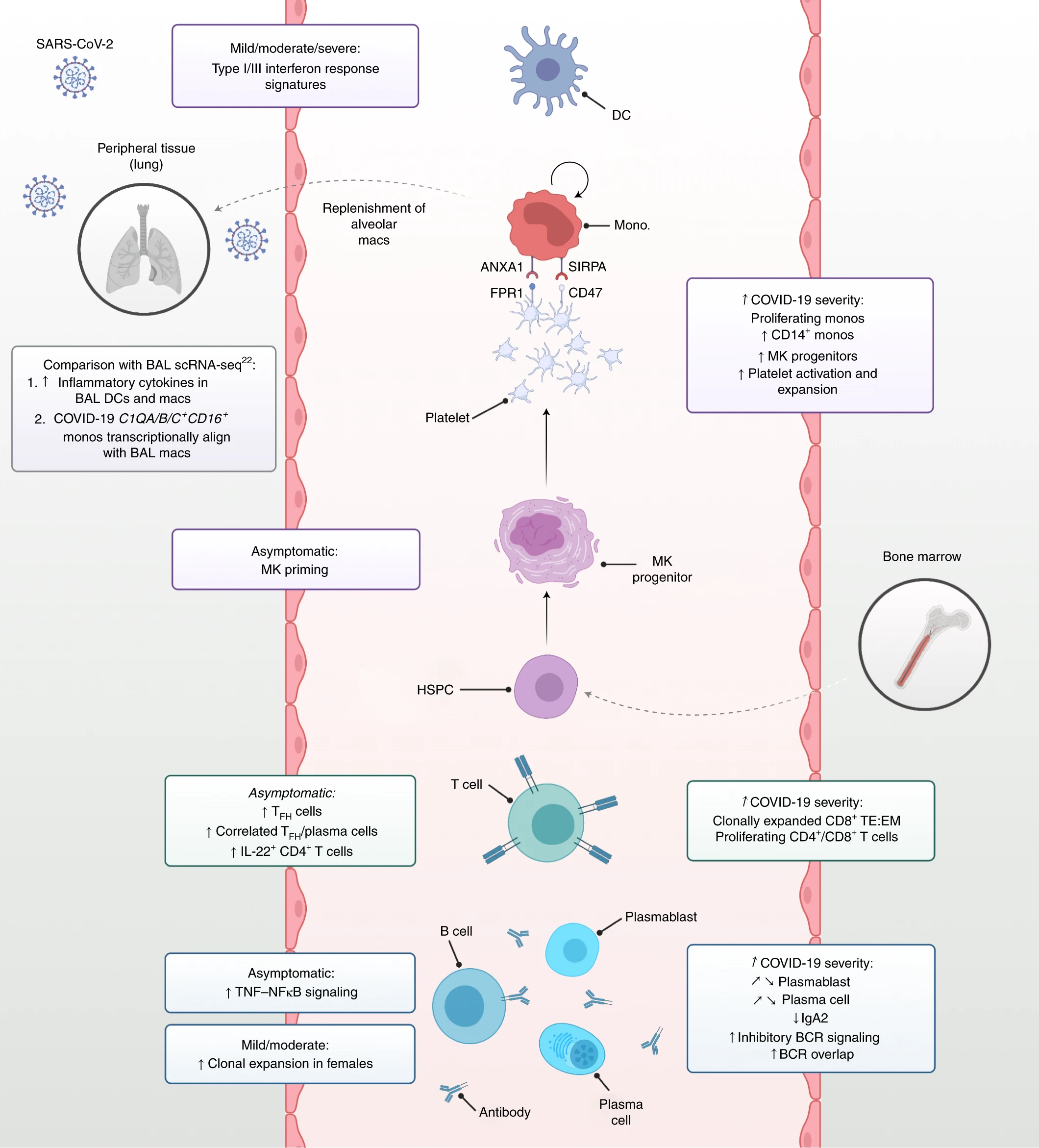
Analysis of human blood immune cells provides insights into the coordinated response to viral infections such as severe acute respiratory syndrome coronavirus 2, which causes coronavirus disease 2019 (COVID-19). We performed single-cell transcriptome, surface proteome and T and B lymphocyte antigen receptor analyses of over 780,000 peripheral blood mononuclear cells from a cross-sectional cohort of 130 patients with varying severities of COVID-19. We identified expansion of nonclassical monocytes expressing complement transcripts (CD16+C1QA/B/C+) that sequester platelets and were predicted to replenish the alveolar macrophage pool in COVID-19. Early, uncommitted CD34+ hematopoietic stem/progenitor cells were primed toward megakaryopoiesis, accompanied by expanded megakaryocyte-committed progenitors and increased platelet activation. Clonally expanded CD8+ T cells and an increased ratio of CD8+ effector T cells to effector memory T cells characterized severe disease, while circulating follicular helper T cells accompanied mild disease. We observed a relative loss of IgA2 in symptomatic disease despite an overall expansion of plasmablasts and plasma cells. Our study highlights the coordinated immune response that contributes to COVID-19 pathogenesis and reveals discrete cellular components that can be targeted for therapy.
|
| Notes |
|